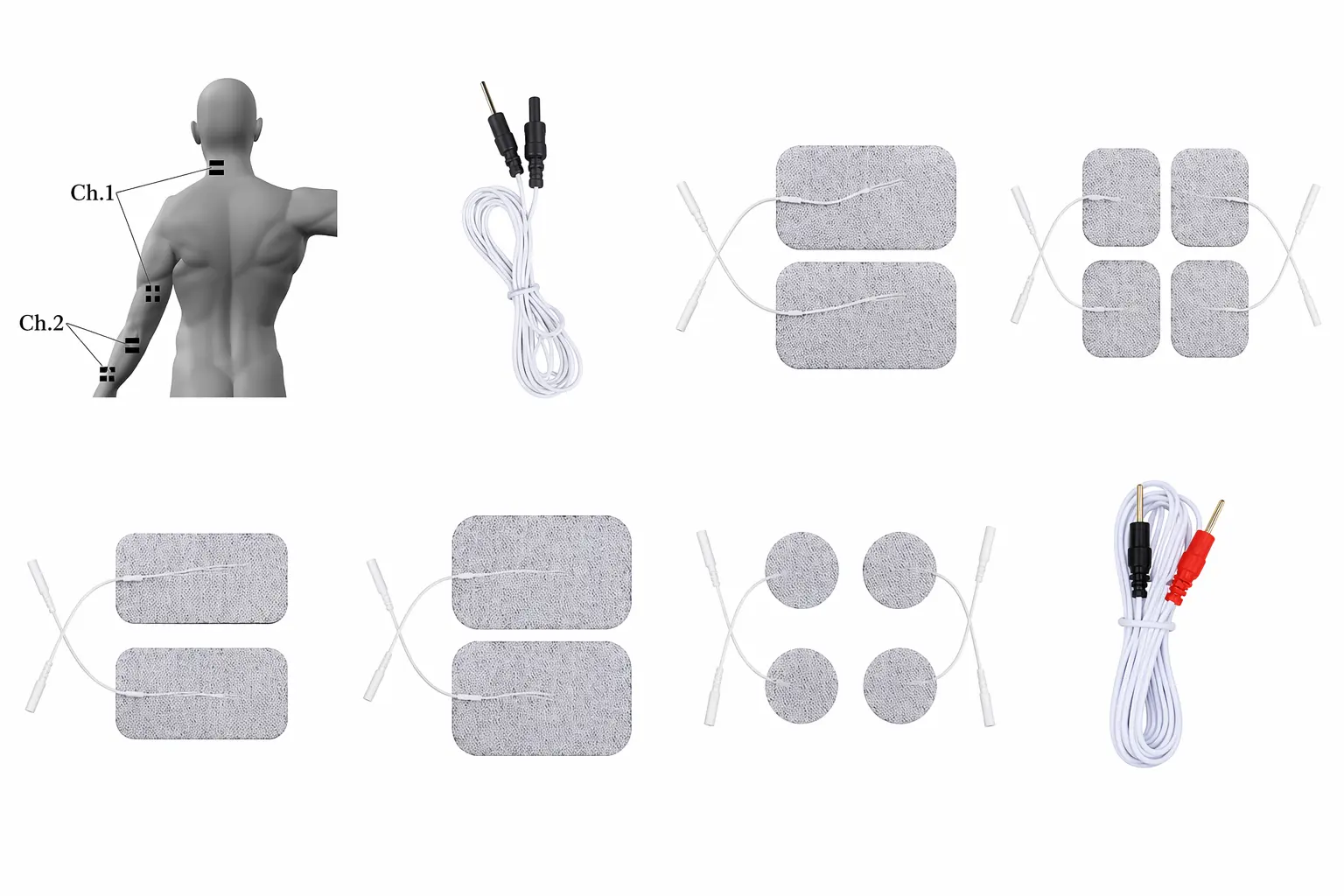
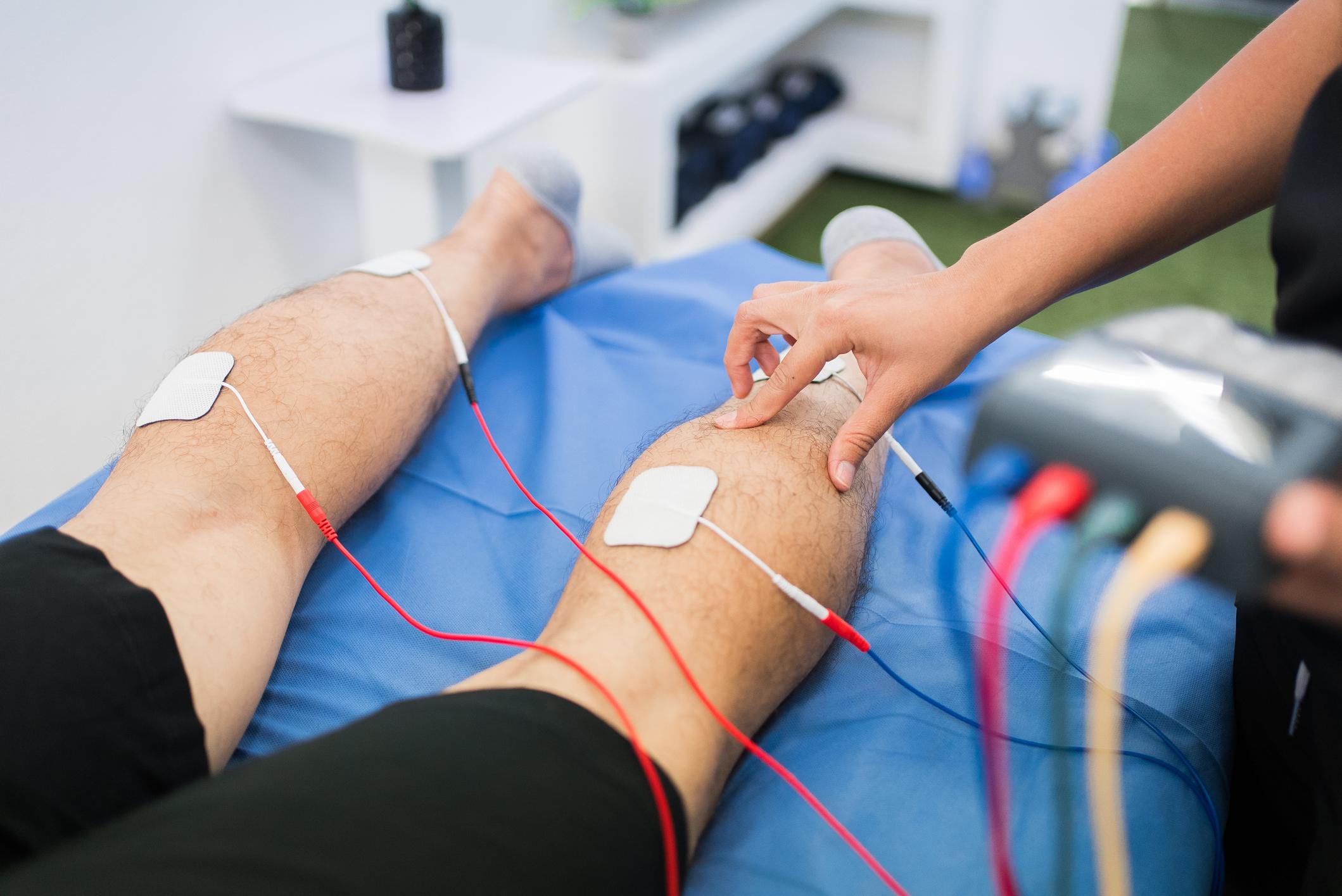
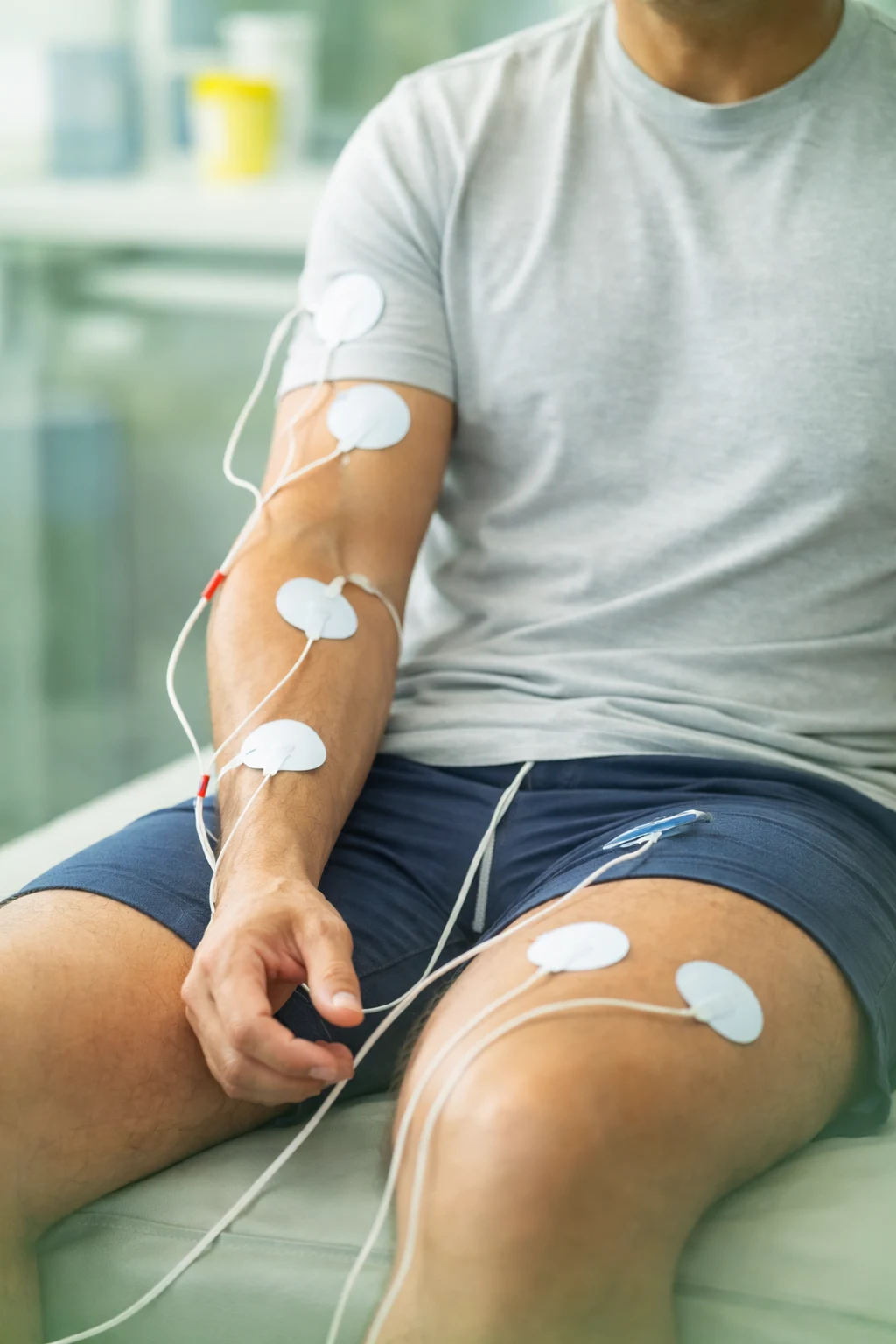
Best Practices for Electrode Placement in Therapy
Correct electrode placement is critical to achieving optimal results in electrotherapy. Whether using TENS, EMS, or EMG devices, positioning the electrodes accurately ensures effective stimulation and patient comfort. Electrodes should be placed over targeted muscle groups or