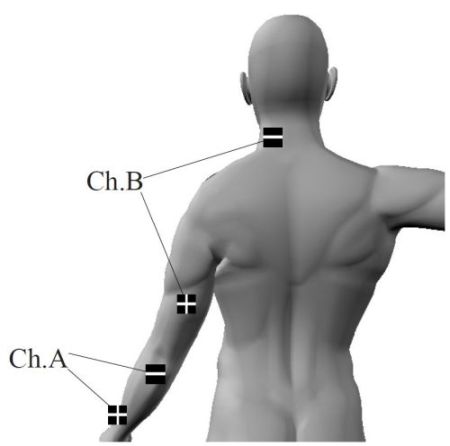
Expected service life: 5 years. Careful use and maintenance extend the life of
the unit over the service life limit.
Environmental Conditions for use: +5 to +40 degrees Centigrade. 15-90%
Humidity. Atmospheric pressure: 700 hPa to 1 060 hPa.
Environmental conditions for storage & transport: -25 to +70 degrees
Centigrade. 15-90% Humidity. Atmospheric pressure: 700 hPa to 1 060 hPa.
Additional requirements for device use.
1 hour required to warm from the minimum storage temperature between uses
until it is ready for intended use.
1 hour required to cool from the maximum storage temperature between uses
until it is ready for intended use.
Physical dimensions: 119.2mm x 69mm x 28.7mm
Weight: 104 g without battery