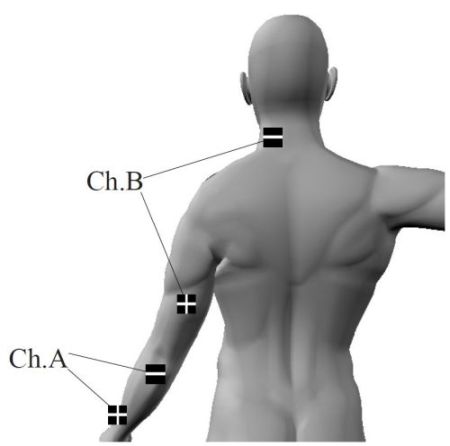
Battery:
Battery set: 4 x 1.5V, AAA battery.
Low battery indication at 4.2-4.4 volts +/- 0.2 volts, automatic shut off when voltage drops below the low indication. Replace the batteries immediately!
The device goes off automatically when not in use (energy saving): for example when in settings and no key is pressed over 1 minute when in stimulation mode (home screen) and all channels are 0mA.
Expected average battery set life [of standard 850 mAh, alkaline]:
14-18 hours in STIM, 28 hours in EMG mode.
Expected service life:
5 years. Careful use and maintenance extend the life of the unit over the service life limit.
Environmental Conditions for use:
+5 to +40 degrees Centigrade. 15-90% Humidity.
Environmental conditions for storage & transport:
-25 to +70 degrees Centigrade. 15-90% Humidity.
Physical:
Dimensions: Length 150 mm, Width 89 mm, Depth 35 mm.